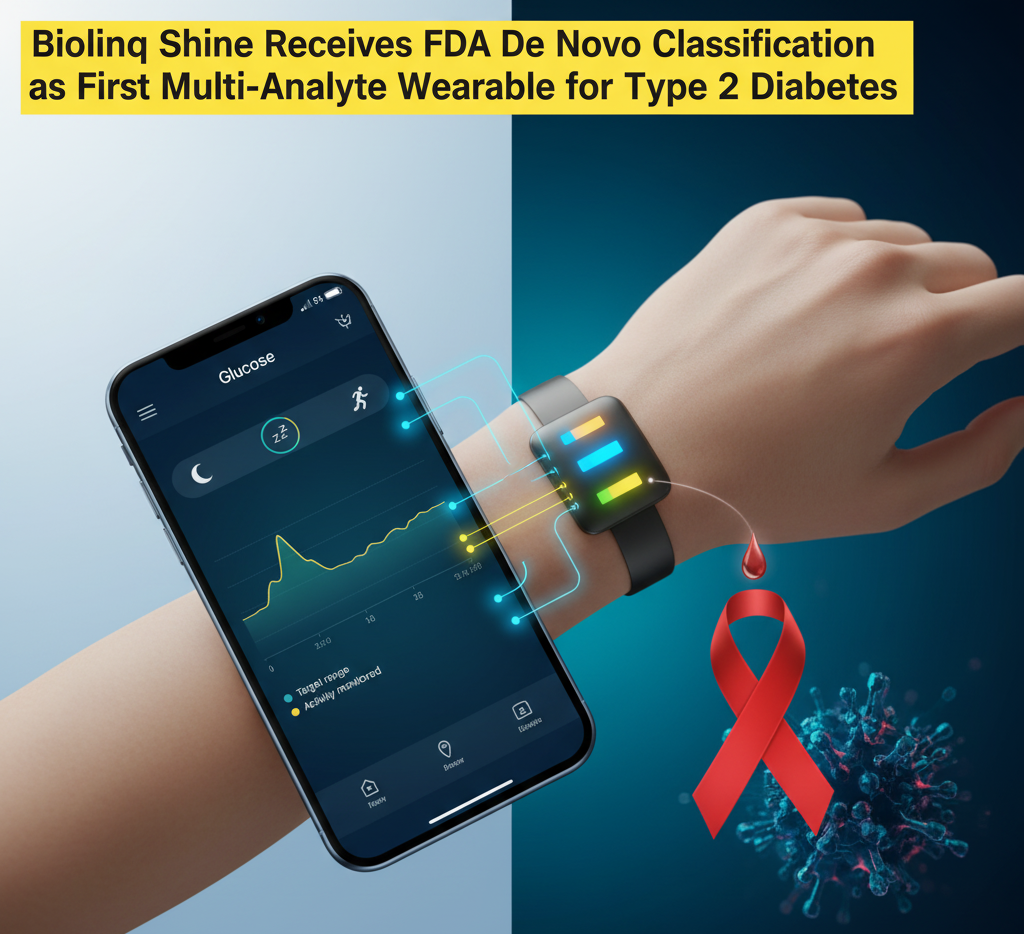
Biolinq Inc. has achieved a significant milestone in diabetes management with the FDA granting De Novo classification to its innovative wearable biosensor, Biolinq Shine. This marks Biolinq Shine as the first fully autonomous, multi-analyte wearable device that integrates real-time glucose, activity, and sleep information into a single, user-friendly device.
The Biolinq Shine biosensor is specifically designed for individuals aged 22 years and older with non-insulin-dependent Type 2 Diabetes (T2D).
Revolutionizing Diabetes Management
Biolinq Shine aims to transform how patients manage their T2D by providing continuous, actionable insights:
- Real-time Glucose Monitoring: Worn as a patch on the forearm, the device provides instant glucose updates through a color-coded LED display. Blue indicates the target range, while yellow signals high glucose levels.
- Integrated Monitoring: Beyond glucose, it simultaneously tracks physical activity and sleep information through an accompanying mobile app.
- Needle-Free Technology: Unlike conventional filament-based continuous glucose sensors, Biolinq Shine does not require a subcutaneous introducer needle for placement. Its microsensor array, built with advanced semiconductor technology, is up to 20 times shallower than typical CGM devices, enhancing comfort and user engagement.
“Biolinq Shine is a first-of-its-kind biosensor designed to support metabolic health for people with diabetes who are not dependent on insulin,” said Dan Bradbury, Chairman of Biolinq. “By automatically tracking glucose levels, physical activity and sleep information, this technology offers meaningful insights that can encourage healthier choices every day.”
Addressing a Critical Unmet Need
Type 2 Diabetes affects a vast population, accounting for 90% to 95% of the over 37 million US adults with diabetes, and impacts nearly 63% of individuals globally. Managing T2D effectively requires maintaining glucose levels within a target range (Time-in-Range or TIR), a goal often difficult to achieve with infrequent, episodic glucose measurements.
Biolinq Shine addresses this by providing continuous data that correlates glucose levels with lifestyle factors, empowering users to make immediate behavioral and lifestyle changes. While the device aids in identifying trends and patterns, Biolinq emphasizes that users should always consult a healthcare provider before taking medical action.
Rich Yang, CEO of Biolinq, expressed enthusiasm for the regulatory achievement: “The entire Biolinq team is thrilled about this regulatory milestone, which is a testament to the outstanding work by our team. We are grateful to the FDA for its rapid and rigorous review in establishing a new category of wearable biosensors.” He also hinted at the broader potential: “We’ve only scratched the surface of what is possible with our multi-analyte-capable biosensor platform in supporting metabolic health for everyone.”
This De Novo classification not only creates a new category for multi-analyte wearable biosensors but also signals a significant leap forward in personalized diabetes management, promising improved metabolic health for millions.







