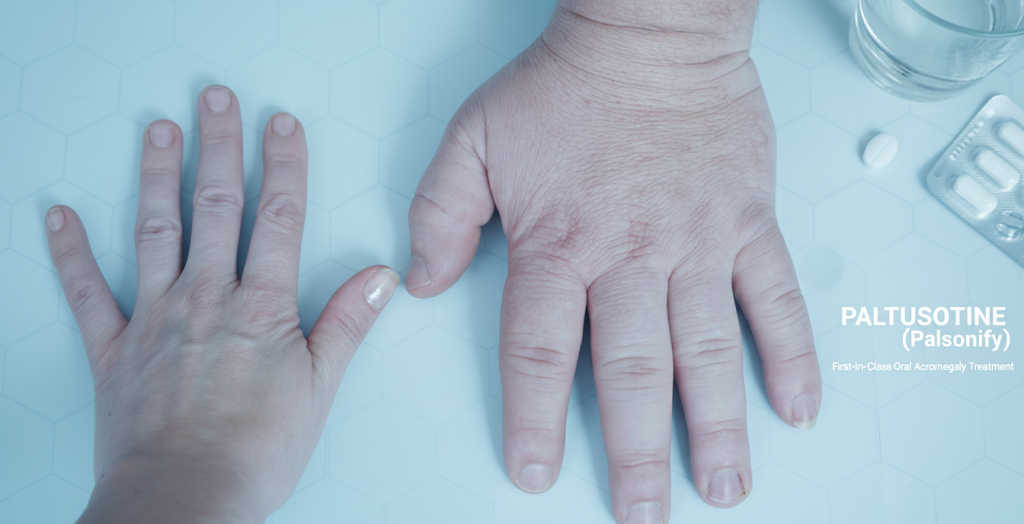
The FDA has approved paltusotine (Palsonify; Crinetics Pharmaceuticals, Inc), marking a historic milestone as the first once-daily, oral treatment for adults with acromegaly. This somatostatin receptor agonist is approved for patients who had an inadequate response to surgery or for whom surgery is not a viable option.
This approval is expected to transform patient care, which previously relied heavily on burdensome injectable therapies. As Jill Sisco, president of Acromegaly Community, stated, the new oral treatment reflects that the patient community’s desire for “maintaining consistent control so the disease doesn’t control us” has been heard. Paltusotine is anticipated to be available in the US in early October 2025.
Understanding Acromegaly
Acromegaly is a rare, chronic condition in adults characterized by the overproduction of growth hormone (GH) by a tumor in the pituitary gland. The excessive GH drives the growth of certain bones, organs, and other tissues.
Symptoms, which often develop slowly and can be difficult to identify early on, include:
- Facial Changes: Thick lips and ears, a broad nose, jutting brow or jaw, and gaps between teeth.
- Enlargements: Enlarged hands, feet, and tongue.
- Skin Changes: Coarse, oily, and thickened skin, acne, and skin tags.
Without effective treatment (which may include surgery and radiation), individuals face a higher risk of serious, life-threatening complications.
Strong Clinical Data Backs Approval
The FDA’s decision is supported by robust data from two pivotal Phase 3 trials: PATHFNDR-1 and PATHFNDR-2. These studies demonstrated paltusotine’s ability to achieve and maintain biochemical control regardless of the patient’s underlying disease severity.
| Clinical Trial | Primary Endpoint Success | Key Findings |
| PATHFNDR-1 | 83% of participants on paltusotine maintained IGF-1 levels at or below the upper limit of normal (vs. 4% on placebo). | Achieved statistical significance on all primary and secondary endpoints. |
| PATHFNDR-2 | 56% of participants on paltusotine achieved IGF-1 levels at or below the upper limit of normal (vs. 5% on placebo). | Demonstrated rapid onset of action and reliable biochemical control. |
Export to Sheets
Furthermore, long-term data from the open-label extension phases confirmed the drug’s ability to deliver durable IGF-1 control, sustained improvements in patient symptom burden, and a consistent safety profile. IGF-1 (insulin-like growth factor 1) is the primary biomarker used to monitor disease activity in acromegaly.







