In a breakthrough for mucosal immunology, researchers at the Massachusetts Institute of Technology (MIT), in collaboration with international partners, have identified a specialized protein that serves as a vital guardian of the human gastrointestinal tract. The protein, known as Intelectin-2 (ITLN2), has been revealed to possess a unique “dual-role” capability: it simultaneously reinforces the gut’s physical defenses and acts as a precision antimicrobial agent against deadly pathogens.
The study, recently published in Nature Communications, offers a promising new roadmap for treating antibiotic-resistant infections and inflammatory conditions by leveraging the body’s own natural defensive biochemistry.
A Multimodal Defender: How It Works
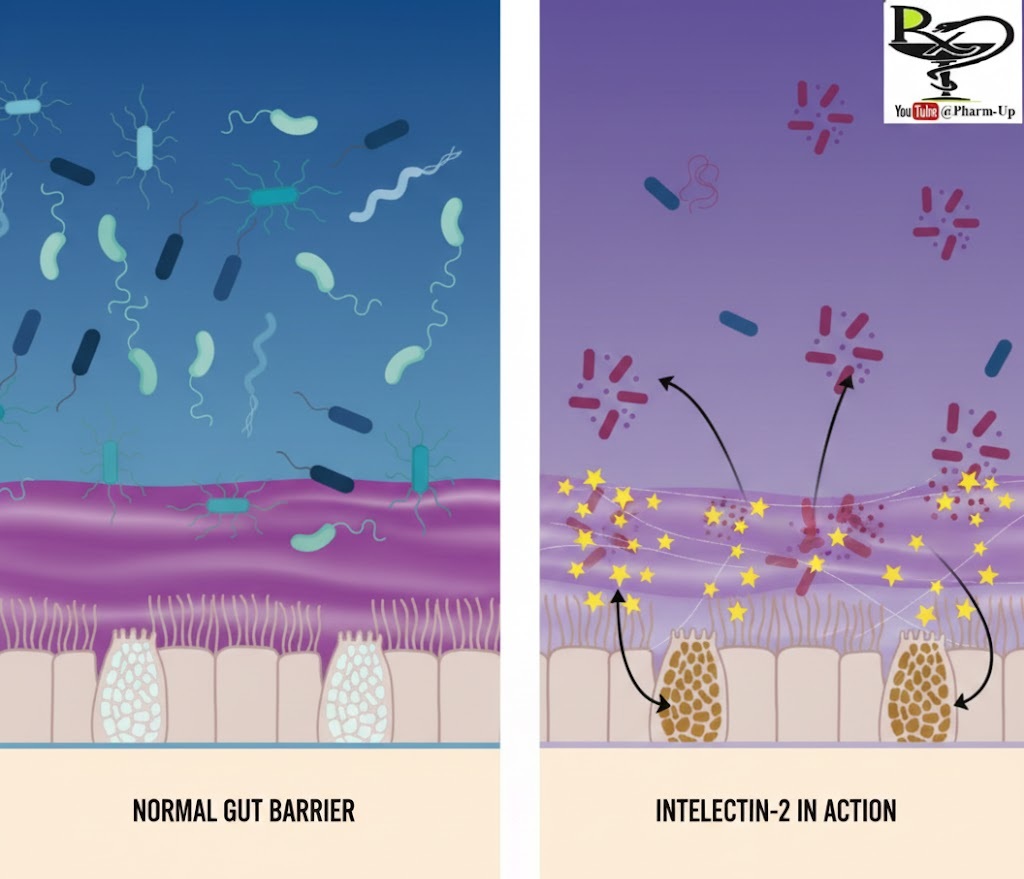
Most protective measures in the body are either passive (like a wall) or active (like a soldier). Intelectin-2 is remarkably both. As a lectin—a protein specialized in recognizing and binding to specific sugar molecules—it monitors the “glycan landscape” of the gut to distinguish between the body’s own protective layers and invading threats.
Phase 1: Strengthening the Physical Wall
The GI tract is lined with mucins, large glycoproteins that form the slippery mucus layer. Intelectin-2 identifies galactose sugars on these mucins. By binding to them, the protein acts as a molecular cross-linker, “stapling” the mucus strands together. This results in a denser, more viscous barrier that makes it physically harder for bacteria to reach the underlying intestinal cells.
Phase 2: Targeted Antimicrobial Assault
When pathogens attempt to penetrate this reinforced wall, Intelectin-2 switches to an offensive mode. It recognizes distinct sugar patterns on the surface of harmful bacteria.
- Sequestration: Once bound, the protein traps the bacteria within the mucus matrix, preventing them from colonizing the gut wall.
- Growth Inhibition: The binding process interferes with the bacteria’s ability to thrive. Over time, the bacteria lose viability, effectively neutralizing the threat without the need for traditional inflammatory immune responses.
A New Weapon Against “Superbugs”
The timing of this discovery is critical. With Antimicrobial Resistance (AMR) becoming a global health crisis, Intelectin-2 offers a mechanism that bacteria find difficult to evade. Because the protein targets fundamental structural sugars on the bacterial surface rather than specific metabolic enzymes (which bacteria can easily mutate), it represents a more “future-proof” defensive strategy.
The research team confirmed that Intelectin-2 is effective against a broad spectrum of microbes, including:
- Staphylococcus aureus: A leading cause of skin and systemic infections, often resistant to multiple drugs (MRSA).
- Klebsiella pneumoniae: A notorious Gram-negative “superbug” frequently found in hospital-acquired infections.
Clinical Implications: Beyond Antibiotics
The discovery opens several doors for future medical therapies:
- Inflammatory Bowel Disease (IBD): Patients with Crohn’s or Colitis often have a “leaky” or compromised mucus layer. Engineering Intelectin-2-based treatments could help “re-seal” the gut.
- Prophylactic Treatments: High-risk patients (such as those undergoing major surgery) could be given Intelectin-2 to bolster their natural barriers against sepsis.
- Microbiome Preservation: Unlike broad-spectrum antibiotics that kill beneficial “good” bacteria, Intelectin-2 appears to be more selective, potentially protecting the gut’s delicate microbial balance.
While further clinical trials are necessary to determine how ITLN2 can be safely administered as a drug, the MIT study provides the foundational proof that the gut’s “passive” barrier is far more dynamic and aggressive than previously believed.